2.4.1.15: alpha,alpha-trehalose-phosphate synthase (UDP-forming)
This is an abbreviated version!
For detailed information about alpha,alpha-trehalose-phosphate synthase (UDP-forming), go to the full flat file.
Reaction
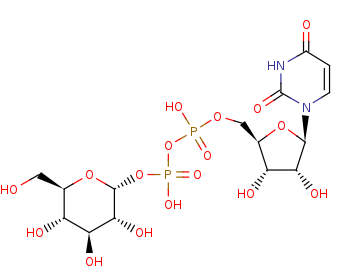 UDP-alpha-D-glucose +
UDP-alpha-D-glucose +
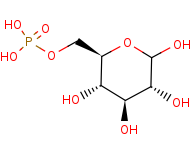 D-glucose 6-phosphate=
D-glucose 6-phosphate=
 UDP +
UDP +
 alpha,alpha-trehalose 6-phosphate
alpha,alpha-trehalose 6-phosphate
Synonyms
alpha,alpha-trehalose phosphate synthase (UDP-forming), alpha,alpha-trehalose-phosphate synthase (UDP-forming), AtTPS1, AtTPS6, GbTPS, glucosyltransferase, uridine diphosphoglucose phosphate, OtsA, OtsA1, OtsA2, phosphotrehalose-uridine diphosphate transglucosylase, T-6-P, T6P synthase, Ta1210, TPS, TPS-2, TPS-3, TPS1, TPS2, TPS3, TPS5, TPSP, transglucosylase, trehalose 6-phosphate synthase, trehalose 6-phosphate synthase 1, trehalose 6-phosphate synthetase, trehalose phosphate synthase, trehalose phosphate synthase 5, trehalose phosphate synthetase, trehalose phosphate-uridine diphosphate glucosyltransferase, trehalose-6-P synthase, trehalose-6-phosphate synthase, trehalose-6-phosphate synthase 1, trehalose-6-phosphate synthase/phosphatase, trehalose-6P synthase, trehalose-P synthetase, trehalosephosphate-UDP glucosyl transferase, UDP-glucose:D-glucose-6-phosphate 1-alpha-D-glucosyltransferase, UDPglucose-glucose-phosphate glucosyltransferase
ECTree
KM Value
KM Value on EC 2.4.1.15 - alpha,alpha-trehalose-phosphate synthase (UDP-forming)
Please wait a moment until all data is loaded. This message will disappear when all data is loaded.
Please wait a moment until the data is sorted. This message will disappear when the data is sorted.
0.6
ADP-glucose
-
37°C, pH 5.5
0.14 - 7.1
D-glucose 6-phosphate
1 - 8.3
glucose 6-phosphate
3.7 - 7
glucose-6-phosphate
0.17 - 1
UDP-alpha-D-glucose
additional information
additional information
-
0.14
D-glucose 6-phosphate

-
at pH 7.4 and 30°C
0.27
D-glucose 6-phosphate
pH 6.0, 60°C, wild-type enzyme
0.34
D-glucose 6-phosphate
pH 6.0, 60°C, N-loop truncation mutant enzyme
0.8
D-glucose 6-phosphate
recombinant enzyme, pH 7.0, 80°C, in presence of Mg2+
3
D-glucose 6-phosphate
-
37°C, pH 5.5, cosubstrate: UDP-glucose
3.9
D-glucose 6-phosphate
-
pH 7.0, 30°C
4.2
D-glucose 6-phosphate
-
37°C, pH 5.5, cosubstrate: ADP-glucose
4.5
D-glucose 6-phosphate
-
pH 8.5, 37°C
5.3
D-glucose 6-phosphate
-
37°C, pH 5.5, cosubstrate: TDP-glucose
7.1
D-glucose 6-phosphate
-
37°C, pH 5.5, cosubstrate: GDP-glucose
0.4
GDP-glucose

-
37°C, pH 5.5
2.17
GDP-glucose
-
pH 8.5, 37°C
1
GDPglucose

-
in absence or presence of a polyanion
1
glucose 6-phosphate

-
reaction with GDPglucose
1.2
glucose 6-phosphate
-
reaction with UDPglucose
2
glucose 6-phosphate
-
-
2
glucose 6-phosphate
-
GDPglucose
3.5
glucose 6-phosphate
-
-
5
glucose 6-phosphate
-
-
7.5
glucose 6-phosphate
-
bifunctional fusion enzyme of trehalose-6-phosphate synthetase and trehalose-6-phosphate phosphatase
8.3
glucose 6-phosphate
-
-
3.7
glucose-6-phosphate

-
-
4
glucose-6-phosphate
-
with GDPglucose as glucosyl donor
7
glucose-6-phosphate
-
with UDPglucose as glucosyl donor
2
TDP-glucose

-
37°C, pH 5.5
2.38
TDP-glucose
-
pH 8.5, 37°C
0.17
UDP-alpha-D-glucose

-
at pH 7.4 and 30°C
0.2 - 1
UDP-alpha-D-glucose
pH 6.0, 60°C, wild-type enzyme
0.74
UDP-alpha-D-glucose
pH 6.0, 60°C, N-loop truncation mutant enzyme
0.3
UDP-glucose

-
37°C, pH 5.5
0.5
UDP-glucose
recombinant enzyme, pH 7.0, 80°C, in presence of Mg2+
2
UDP-glucose
-
pH 8.5, 37°C
9.6
UDP-glucose
-
pH 7.0, 30°C
0.04
UDPglucose

-
in presence of optimal concentration of a polyanion
5.1
UDPglucose
-
bifunctional fusion enzyme of trehalose-6-phosphate synthetase and trehalose-6-phosphate phosphatase
additional information
additional information

-
-
-
additional information
additional information
-
-
-
additional information
additional information
-
bifunctional fusion enzyme of trehalose-6-phosphate synthetase and trehalose-6-phosphate phosphatase
-
additional information
additional information
-
complex bimodal kinetics
-
additional information
additional information
the high catalytic efficiency of OtsA results from the high affinity of the enzyme for uridine 5'-diphosphoglucose at low temperatures
-
additional information
additional information
-
the high catalytic efficiency of OtsA results from the high affinity of the enzyme for uridine 5'-diphosphoglucose at low temperatures
-




 results (
results ( results (
results ( top
top






